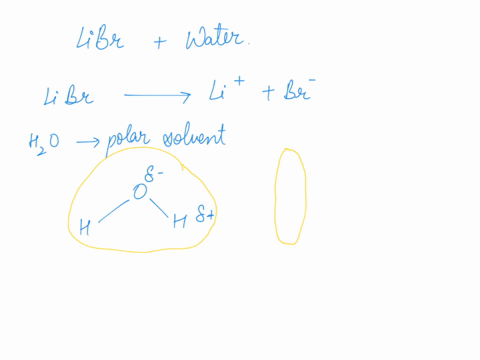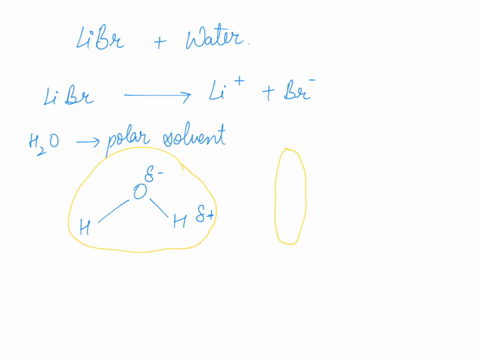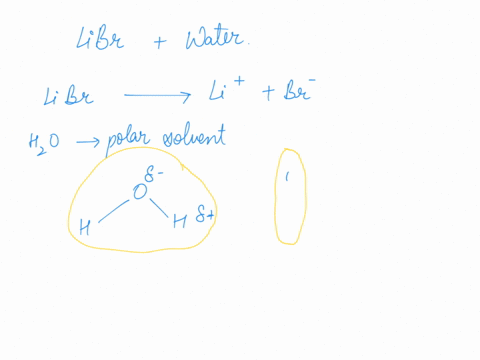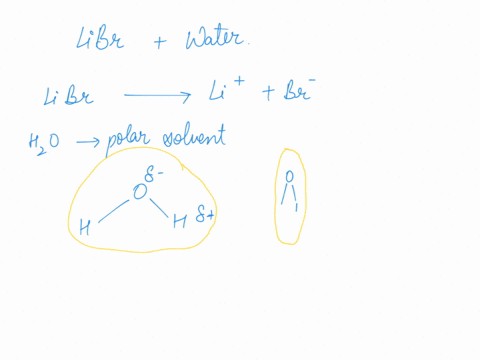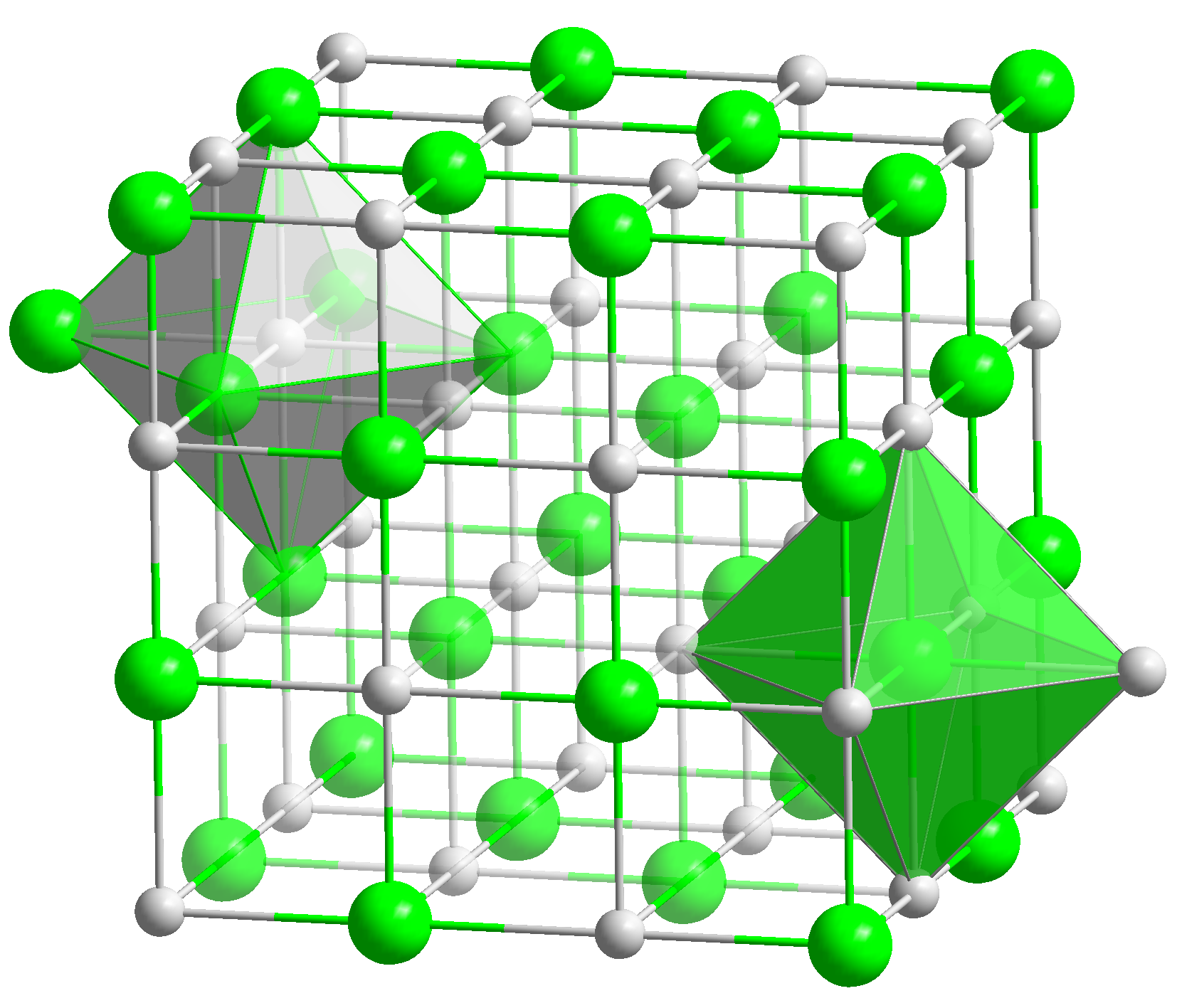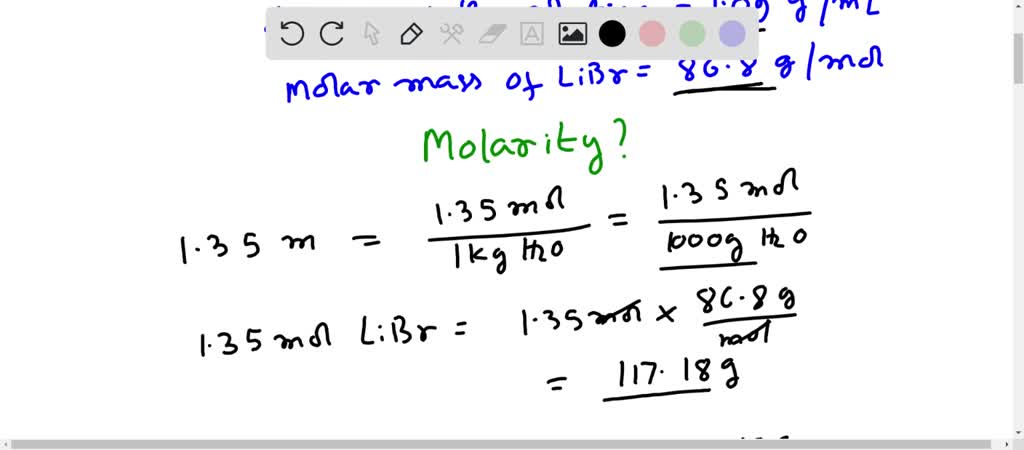
SOLVED: An aqueous solution of LiBr (molar mass = 86.8 g/mol) has a molality of 1.35 m and a density of 1.09 g/mL (1 kg of water). What is its molarity?

Libr Refrigerant Lithium Bromide 55% Solution With Molybdate Inhibited, For Industrial at best price in Srirangam

Metals | Free Full-Text | Corrosion Behavior of Steels in LiBr–H2O–CaCl2–LiNO3 Systems
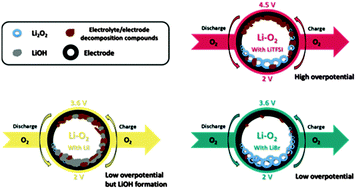
Li–O2 cells with LiBr as an electrolyte and a redox mediator - Energy & Environmental Science (RSC Publishing)

Stable Phase Equilibria in the Quaternary Systems LiBr–NaBr–SrBr2–H2O and LiBr–KBr–SrBr2–H2O at 288.15 K | Journal of Chemical & Engineering Data